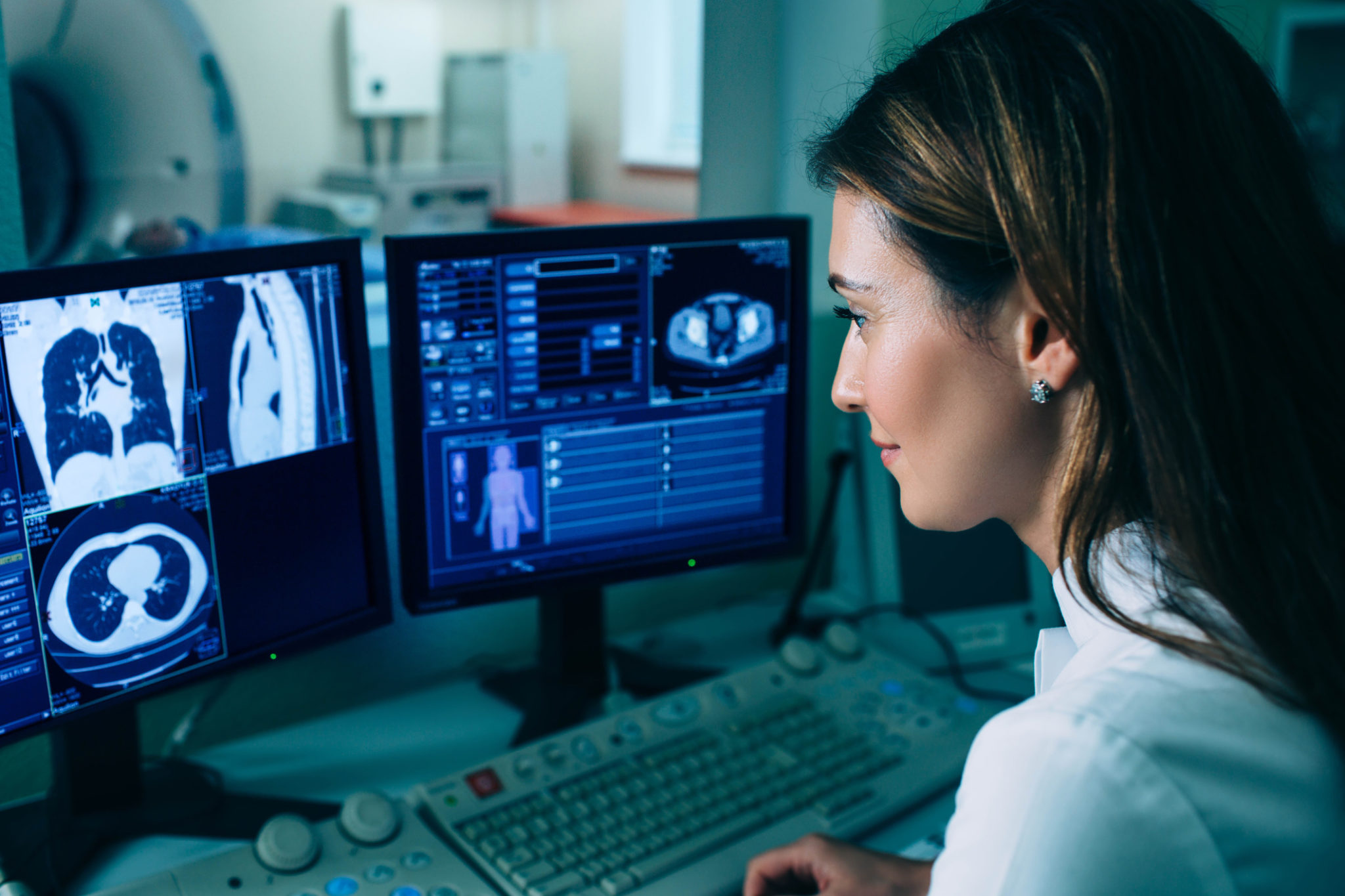
Imaging studies are an important part of screening and diagnosis for some cancers, lung, and breast in particular. Such studies have led to more lung and breast cancers being diagnosed at a smaller size compared to what was found prior to the advent of screening programs. One important research question that is currently being explored is whether the use of artificial intelligence to aid in diagnosis can improve the performance of radiologists alone. Let’s take a look at what we know so far.
Breast cancer screening
According to the American Cancer Society (ACS), approximately one in eight women will be diagnosed with breast cancer in their lifetime. It is the second leading cause of death from cancer in women.
Breast cancer screening is commonly performed on patients who have no obvious signs of disease. Many of these women are not at high risk for the disease. Nor do they have a family history.
Although many preventive health guidelines recommend screening mammograms, concerns have been raised. For example, one in five abnormal mammograms is a false positive. That means the mammogram was read as positive by a radiologist but proved not to be cancer on biopsy.
Over the span of ten years, about half of women are given a false-positive result. This usually leads to further testing, anxiety, distress, and sometimes unnecessary procedures or treatment.
The Google Health study
Experts from Google Health and its subsidiary, Alphabet’s DeepMind unit, recently worked with Northwestern University, Cancer Research UK Imperial Center, and Royal Surrey County Hospital to examine aspects of radiographic breast cancer diagnosis. In particular, they wanted to better understand the reasons for inaccuracies in the diagnosis of breast cancer. And, they wanted to determine if artificial intelligence could help.
In order to comprehend how AI can be used to improve the results of breast imaging moving forward, it is important to have a basic understanding of how this artificial intelligence system works. This is a type of system known as “Deep Learning” which involves a three-dimensional model:
- The first component is analyzing of imaging with nodules already confirmed as cancerous in order to obtain “training data”.
- The second component is training the system to detect a “region of interest” that is suspicious.
- Finally, the third step involves developing a cancer prediction model that would operate independently of the first two steps.
International evaluation of an AI system for breast cancer screening
The results of this research were recently published in the journal Nature in an article titled “International evaluation of an AI system for breast cancer screening.” The study compared the results of mammography readings in an artificial intelligence model to those read by radiologists. There were close to 26,000 women from the UK and over 3,000 women in the United States in the study.
The researchers found that the artificial intelligence model reduced both false positives (when patients are told they have cancer when they don’t) and false negatives (when the disease is present, but not diagnosed).
Although in this early testing the AI caught cancers missed by radiologists, there were also cases in which it missed cancer that was caught by radiologists. This suggests that AI alone may not be the sole solution moving forward.
Screening for lung cancer with low-dose chest CT
With approximately 160,000 deaths in 2018 due to lung cancer, it is the most common cause of cancer death in the United States. The U.S Preventive Services Task Force’s (USPSTF) new guidelines for the use of low dose computed tomography has recently been updated for individuals at high risk of having lung cancer.
Lung cancer screening using this type of computed tomography testing has been shown to reduce death by 20-40%. However, similar to breast cancer screening, one ongoing issue with the use of this screening exam has been the high rate of false positives (a result that indicates that a person has a disease when they actually do not). Although low-dose lung CTs have helped immensely in early detection, it has been found that about one-quarter of the suspected nodules are actually not cancerous.
To determine if this could be improved upon, doctors at Northwestern University and Stanford, teamed up with Google to determine if the same type of artificial intelligence, called Deep Learning, could help improve upon our current methods with lung cancer.
The lung cancer screening AI study
Researchers from Google used more than 42,000 CT scans to train this artificial intelligence system to detect cancerous lung nodules on radiology imaging. The study, titled “End-to-end lung cancer screening with three-dimensional deep learning on low dose chest computed tomography” was published in Nature as well.
Over 6,000 National Lung Cancer Screening Trial cases were tested in this study. In addition, there was an independent evaluation of a set of over a thousand cases. The performance of the artificial intelligence system was compared against radiologists who had evaluated low-dose chest computed tomography scans for patients – several of which had confirmation of cancer by biopsy within a year.
This deep-learning artificial intelligence system produced fewer false negatives (a result that indicates that a person does not have a disease when they actually do) as well as fewer false positives. When prior imaging was available, the model performed better than the radiologists (six of them) with an 11% reduction in false positives and a 5% reduction in false negatives.
What’s next in AI-assisted lung-cancer screening?
The Nature study was a retrospective study that examined past cases. This type of study design is not as strong as prospective studies with randomization. Mozziyar Etemadi, MD, Ph.D., one of the authors of the study has said that “the next step is to perform a prospective study to see if the tool, when used by a radiologist, can lead to earlier and more accurate diagnosis of cancer”.
Another caveat is that it may be some time before AI with deep learning is routinely used in hospital and free-standing radiology suites. The algorithm that is the backbone of the AI-deep learning system is very sophisticated and will undoubtedly require some painstaking work to fully integrate into hospital computer systems. Further, the variability of many cancers could make new scenarios difficult for the deep learning system to interpret if they have not been seen before.
We also need to consider that although AI with deep learning improves some aspects of cancer screening diagnoses, it is not (yet) perfect. It may be that the best way to introduce AI into imaging analysis is to add it to the workflow of radiologists. This is because both have the potential to not catch something or make mistakes.
The bottom line
The performance of the deep learning system shows that there can be a beneficial role of artificial intelligence in cancer screening moving forward. In fact, the use of algorithms that incorporate co-morbidities and risk factors in medicine is not uncommon today. However, the use of such a sophisticated one on its own will most certainly take time. It will also require well-designed prospective studies that follow patients over time. Nonetheless, there is no denying that there will be an important role of artificial intelligence in cancer screening moving forward.
Related Content: Google, Big Data, and Public Health
More Articles by This Author:
CAR T-Cell Therapy for Cancer: What’s New on the Road to Cure?
Research: New Laser Tech Can Detect Circulating Cancer Cells
***
Joshua Mansour, M.D.
Website:
https://www.doctorjoshmd.com/
Dr. Joshua Mansour received his undergraduate degree from Vanderbilt University where he was a chancellor scholar. He then went on to Ross University School of Medicine to continue his education.
Dr. Mansour recently received Ross University School of Medicine 2019 Early Distinguished Career Award, given to a “young alumni” that has already experienced a high level of success and displayed promise for future growth and leadership opportunities.
He completed his internal medicine residency at Medical College of Georgia, completed a fellowship in Hematology and Oncology at the Medical University of South Carolina, and then went to Stanford University to pursue an additional fellowship in Bone Marrow Transplantation and Cellular Immunotherapy. He is triple board-certified in internal medicine, hematology, and oncology.
Dr. Mansour actively belongs to several associations including the Medical Oncology Association of Southern California, where he is currently a board member, the California Medical Association, American Society of Clinical Oncology, the Community Oncology Association, where he is on the CAR T-Cell Task Force, American Society of Hematology, and American Society of Transplantation & Cellular therapy. He also serves as a physician mentor for students as part of the American College of Physicians.
Although he has an extensive and varied medical background and training, he currently focuses on hematological malignancies. He is a bone marrow transplant and cellular immunotherapy physician in Los Angeles, CA. This is where he sees patients, continues research in this field, as well as helping with clinical trials.
He dedicates much of his spare time investigating ways to improve patient care and outcomes. He has helped design and implement clinical studies to evaluate current treatment plans, collaborated on grant proposals, and lead multi-institutional retrospective studies that have been published in major high impact medical and scientific journals.
In addition to these studies, he has been writing for several years. His commentaries and pieces have been published in several outlets including US News & World Report, Cancer Therapy Advisor, DrugWatch, Today’s Practioner, KevinMD, and Physician’s News. Dr. Mansour has also been featured on FOX television among other media outlets.
Most recently he was the recipient of the “40 under 40 in Cancer” Award. This award is given to recognize the contributions being made across the field of cancer by rising stars and emerging leaders under the age of 40. Recipients are selected by a panel of reviewers from across diverse roles in oncology.
Comments:
Leave a Reply
Comment will held for moderation











Let’s not forget the real big picture behind the large global mass media dissemination blast of this so-called study. Mammograms should be abolished for NUMEROUS scientifically solid reasons despite the MISLEADING concept of better diagnosis pushed by the highly profiteering corrupt corporate medical industry (which google is part of because it founded “Calico”, is a stakeholder in “One Medical”, etc) — read the books: ‘Mammography Screening: Truth, Lies and Controversy’ by Peter Gotzsche and ‘The Mammogram Myth’ by Rolf Hefti
I think several people, myself included, have witnessed first hand how mammograms have saved the lives of thousands through early detection for breast cancer, in addition to low dose CT scan screening for lung cancer. These screening tests should not be abolished. I will look in to these books though, thank you for the recommendations.